
Why Evolutionary Populations?
Biodiversity in general, and agrobiodiversity in particular, are important to humans for several reasons: they increase resilience to climate changes by increasing the stability of ecosystem processes [1], benefit health through dietary diversity [2], are important for food security [3] and reduce the risk of yield losses [4].
However, biodiversity in general, and crop biodiversity in particular, are threatened and plant breeding is thought of being one of the causes [5]. The decline in agrobiodiversity is due to the predominant philosophy being followed by both public and private breeders with regard to two fundamental concepts, namely, wide adaptation, defined as the ability of a variety to perform well across locations, and stability, defined as yield consistency across years [6–8]. In fact, despite the evidence from evolutionary biology that locally adapted plants perform better [9] and demonstrate an average 45% higher fitness than introduced genotypes [10], selection for specific adaptation has been rather an exception in plant breeding and only a few studies have measured its impact [11].
Moreover, plant breeding has followed the changes in global food systems shifting towards a reduced number of crops [12], with markets’ preference for uniformity and standardization [13], thus becoming one of the major drivers of climate change, land-use change and biodiversity loss [14]. In other words, it is the requirements of industrial cultivation, husbandry and processing (and to some extent consumer demand) that determine the breeding objectives rather than nutritional value, taste, improved stress resistance or adaptation to natural conditions [15]. Related to this scenario is the evidence that global agriculture has grown more vulnerable to climate change [16].
One of the effects of climatic changes on agriculture is in fact a decline in resilience of crop systems, as recently suggested for wheat in Europe [17]. The same authors argue that cultivating “a set of varieties with diverse responses to critical weather conditions is required to promote the climate resilience of a crop”. This approach, which uses agro-biodiversity in the form of crop heterogeneity to achieve food security, has already proven its effectiveness in reducing powdery mildew in barley [18] and in reducing rice blast thereby increasing grain yield in China [19].
In order to cope with the complex and combined effects of challenges such as human population increase, food security, food safety and human health, we need to reconsider the role of diversity in plant breeding. One strategy, which has proved to be efficient in maximizing genetic gains while deploying crop diversity has been decentralized selection, defined as selection in the target environment(s) [20,21]. With the emphasis on specific adaptation, decentralized selection differs from centralized breeding by adapting crops to each target environment through different, locally adapted, varieties. Decentralized selection recognizes the importance of keeping genotype ´ location (GL) interaction distinct from genotype ´ year (GY) interaction, as already recommended more than 50 years ago [22, 23]. By subdividing the target population of environments in sub groups, each characterized by repeatable GL interaction, we can exploit genotype ´ environment interactions (GEI). In fact, in this way, the variance component due to GEI is minimized, and therefore both heritability and selection gains are maximized [24]. However, a distinction is rarely made in the literature between GL and GY [25]. This is unfortunate because GY is largely unpredictable while GL can to some extent be repeatable and therefore predictable. Moreover, decentralized selection can make a positive use of GL interactions by selecting for specific adaptation, while the solution to GY are well buffered cultivars against unpredictable fluctuations of the environment, which are increasing in frequency and extent as a consequence of climate change [26, 27]. The buffering against environmental fluctuations can be achieved by growing heterogeneous populations, which combine both individual and population buffering. While individual buffering is a property of specific genotypes, and particularly of heterozygotes, population buffering arises by the interactions among the different genotypes within a population, beyond the individual buffering of specific genotypes; heterogeneous populations have the advantage of exploiting both individual and population buffering [22, 23].
Evolutionary populations are the heterogenous populations that more than 50 years ago were considered as the ideal solution to environmental fluctuations as those that we are experiencing today. It is interesting that the term “heterogenous” used then, reappeared in the new European Regulation of Organic Agriculture, which will allow the commercialization of seed of “Organic Heterogenous Material”.
References
- Loreau M, de Mazancourt C. 2013. Biodiversity and ecosystem stability: a synthesis of underlying mechanisms. Ecology letters 16: 106 -115
- Singh RK, Chang HW, Yan D, Lee KM, Ucmak D, Wong K, Abrouk M, Farahnik B, Nakamura M, Zhu TH, Bhutani T, Liao W, 2017. Influence of diet on the gut microbiome and implications for human health. Journal of Translational Medicine 15(1):73
- Zimmerer KS, de Haan S. 2017. Agrobiodiversity and a sustainable food future. Nature Plants 3: 17047
- Renard D, Tilman D. 2019. National food production stabilized by crop diversity. Nature 571: 257 – 260
- van der Wouw M, Kik C, van Hintum T, van Treuren R, Visser B. 2010. Genetic erosion in crops: concept, research results and challenges. Plant Genetic resources: characterization and utilization 8 (1): 1-15
- Barah BC, Binswanger HP, Rana BS, Rao NGP. 1981.The use of risk aversion in plant breeding; Concept and application. Euphytica 30: 451–458.
- Lin CS, Binns MR. 1988. A method of analysing cultivar x location x year experiments: A new stability parameter. Theor. Appl. Genet. 76 : 425–430.
- Evans LT. 1993. Crop Evolution, Adaptation and Yield; Cambridge University Press: New York, NY, USA.
- Leimu R, Fischer M. 2008. A Meta-Analysis of Local Adaptation in Plants. PLoS ONE 3 : e4010.
- Hereford J. 2009. A Quantitative Survey of Local Adaptation and Fitness Trade-Offs. Am. Nat. 173: 579–588.
- Kissing Kucek L, Dawson JC, Darby H, Mallory E, Davis M, Sorrells ME. 2021. Breeding wheat for weed-competitive ability: II–measuring gains from selection and local adaptation. Euphytica 217: 203.
- Mulvany P. 2021. Sustaining agricultural biodiversity and heterogeneous seeds. In Rethinking Food and Agriculture. Woodhead Publishing Series in Food Science, Technology and Nutrition; Kassam, A., Kassam, L., Eds.; Woodhead Publishing: Swanston, Cambridge, UK. pp. 285–321.
- Chaudhary P, Bhatta S, Aryal KP, Joshi BK. 2021. Threats, drivers and conservation imperative of agrobiodiversity. J. Agric. Environ. 21: 44–61.
- Springmann M, Clark M, Mason-D’Croz D, Wiebe K, Bodirsky BL, Lassaletta L, de Vries W. Vermeulen SJ, Herrero M, Carlson KM, et al. 2018. Options for keeping the food system within environmental limits. Nature 562: 519–525.
- Wolff F. 2003. Industrial Transformation and Agriculture: Agrobiodiversity Loss as Sustainability Problem. In Governance for Industrial Transformation, Proceedings of the 2003 Berlin Conference on the Human Dimensions of Global Environmental Change; Klaus J, Manfred B, Anna W, Eds.; Environmental Policy Research Centre: Berlin, Germany, 2004 pp. 338–355.
- Ortiz-Bobea A, Ault TR, Carrillo CM, Chambers RG, Lobell DB. 2021. Anthropogenic climate change has slowed global agricultural productivity growth. Nat. Clim. Chang. 11: 306–312.
- Kahiluoto H, Kaseva J, Balek J, Olesen JE, Ruiz-Ramos M, Gobin A, Kersebaum KC, Takáč J, Ruget F, Ferrise R, Bezak P, Capellades G. et al. 2019. Decline in climate resilience of European wheat. Proc. Natl. Acad. Sci. (USA) 116 (1): 123-128
- Wolfe MS, Ceccarelli S. 2020. The need to use more diversity in cereal cropping requires more descriptive precision. Journal of the Science of Food and Agriculture 100: 4119–4123.
- Zhu Y, Chen H, Fan J, Wang Y, Li Y, Chen J, Fan J-X, Yang S, Hu L, Leung H, Mew TW, Teng TW. et al. 2000. Genetic diversity and disease control in rice. Nature 406: 718–722.
- Ceccarelli S. 2015. Efficiency of plant breeding. Crop Science 55: 87-97.
- Ceccarelli S, Grando S, 2022. Return to agrobiodiversity: participatory plant breeding. Diversity 14:126 https://doi.org/10.3390/d14020126.
- Allard RW, Hansche PE. 1964. Some parameters of population variability and their implications in plant breeding. Advances in Agronomy 16: 281‒325.
- Allard RW, Bradshaw AD. 1964. Implications of Genotype-Environmental Interactions in Applied Plant Breeding. Crop Science 4: 503-508.
- Cobb JN, Juma RU, Biswas PS, Arbelaez JD, Rutkoski J, Atlin G, Hagen T, Quinn M, Ng EH. 2019. Enhancing the rate of genetic gain in public-sector plant breeding programs: Lessons from the breeder’s equation. Theoretical and Applied Genetics 132: 627-645.
- Arief VN, DeLacy IH, Basford KE. 2020. Design and analysis of multi-year field trials for annual crops. In (Kang, M.S. ed). Quantitative genetics, genomics and plant breeding. Wallingford, UK, pp 178-193
- Baethgen WE. 2010. Climate Risk Management for Adaptation to Climate Variability and Change. Crop Science 50: S-70–S-76
- Thornton PK, Ericksen PJ, Herrero M, Challinor AJ. 2014. Climate variability and vulnerability to climate change: a review. Global Change Biology 20: 3313–3328

Evolutionary Populations and Mixture
The first research on mixtures and populations dates back to almost a century ago [1]. Over time, different terms such as bulks, evolutionary populations, populations, composite crosses and mixtures have been used. The difference between these terms has recently been clarified [2] and the main difference is between bulks, evolutionary populations, populations, composite crosses on the one hand and mixtures on the other.
Definitions
Mixtures
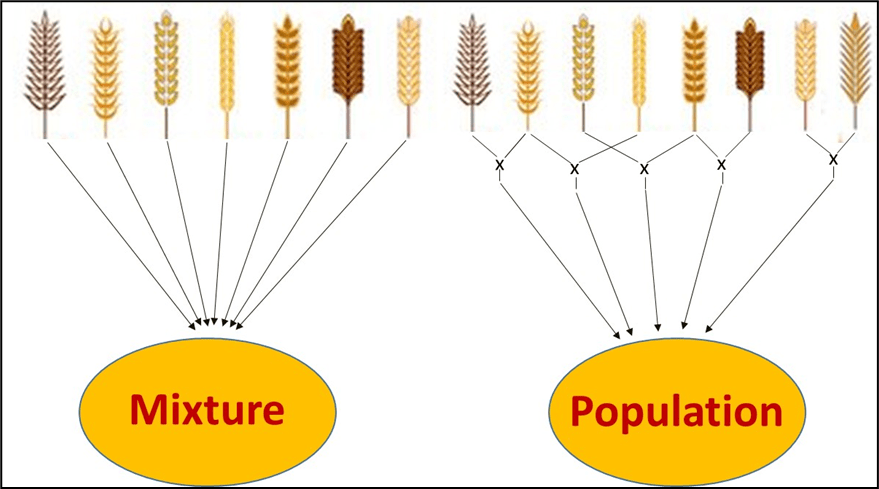
A mixture is obtained by mixing a given quantity (generally a given number) of seeds of different varieties of the crop in question (Figure 1, left).
A mixture can be static or dynamic: a static mixture is composed by mixing a certain number of seeds of each component at the beginning of each season so that the composition of the mixture does not change over time. They are static because, although such mixtures are genetically more complex than monocultures and may therefore be subject to natural selection, they do not capture the effects of natural selection that occurs in the field.
A well-known example of a static mixture is the mixture, in this case of two different crops, barley and wheat, common in some areas of Ethiopia and Eritrea, known as hanfets. Farmers usually mix 60-65% of wheat, which tillers less than barley, and 35-40% of barley. One of the reasons farmers grow the mixture is to stabilize yields across seasons because barley is more drought tolerant than wheat, while wheat is more productive than barley in wet years. Therefore, it is not convenient to let the mixture evolve because, for example, after a dry year the composition of the mixture will be modified in favour of barley: if part of the harvested seed is planted in the following year and the season turn out to be wet, the amount of wheat left in the hanfets may be less than the optimum to fully exploit the wet season [3].
A research conducted in Eritrea for three cropping seasons has confirmed that hanfets yield more that the wheat component and are more stable that the pure wheat and barley [4].
A dynamic mixture is a mixture in which the seed produced in a given year is used as the seed for the next crop, thus capturing the effects of natural selection.
Populations
A population is obtained by mixing the seed obtained by crossing a certain number of varieties in all possible combinations (Figure 1, right). Each year, the population is propagated using the seed produced the year before. The populations have been called bulks, Composite Cross, Composites or evolutionary populations. This last term tells us that these populations, due natural crosses, genetic recombination and natural selection, evolve, adapting better and better to the environment (physical and agronomic) in which they are grown.
The distinction between mixtures and populations is important because:
a) Static mixtures do not evolve;
b) Dynamic mixtures evolve, but not as fast as populations due to their initial genetic composition (mixtures of more or less uniform varieties);
c) Populations evolve faster than dynamic mixtures because they start from highly heterogeneous material (segregating populations derived from crosses).
Figure 1. The difference between a mixture and a population: a mixture is obtained by mixing seed of different varieties while a population is obtained by mixing the seed obtained by crossing different varieties. A mixture can be static or dynamic [3].
References
- Harlan HV, Martini ML. 1929. A composite hybrid mixture. J. American Society of Agronomy 21: 487–490
- Wolfe MS, Ceccarelli S. 2020. The need to use more diversity in cereal cropping requires more descriptive precision. Journal of the Science of Food and Agriculture 100: 4119–4123
- Ceccarelli S, Grando S. 2022. Evolutionary Plant Breeding with an introduction to Participatory Plant Breeding, The Alliance of Bioversity International and CIAT, Rome pp 125 (in press).
- Woldeamlak A, Grando S, Maatougui M, Ceccarelli S. 2008. Hanfets, a barley and wheat mixture in Eritrea: yield, stability and farmer preferences. Field Crops Research 109. 50 – 56.

The Science of Mixtures and Evolutionary Populations
A wealth of genetic, ecological and evolutionary research, spanning over several decades has shown that reincorporating diversity into agro-systems to promote ecosystem services is one viable approach for reducing environmental impact while maintaining and even increasing yields [1].
This large wealth of research has been recently reviewed [2] and has shown that:
1. Evolutionary populations and mixtures are able to adapt their phenology to the location in which they are multiplied.
2. They evolve becoming more and more productive.
3. Evolutionary populations, and to a lesser extent mixtures, have a more stable yield over time than uniform varieties but not over space, i.e., they become specifically adapted.
4. Evolutionary populations and mixtures evolve, becoming more and more resistant to diseases.
5. Evolutionary populations and mixtures control weeds better then uniform varieties even though the scientific evidence on this advantage is still limited.
From all these demonstrated advantages it is possible to extrapolate that evolutionary populations and mixtures are able to slowly adapt to climate change as long as they have, and are able to maintain, a large genetic diversity, and therefore the way they are managed by farmers is important. Evolutionary populations and mixtures, with their capacity to evolve in response to both biotic and abiotic stresses, are likely to be the quickest, cost-effective and evolving solution to such a complex and evolving problem as climate change, with the additional advantage of increasing yield gains resulting from a combination of natural and artificial selection [3, 4, 5].
The speed of adaptation depends also on the type of selection.
Several field studies of natural populations of plants and animals have shown abundant evidence for directional selection on morphology and life history traits (number, size and sex ratio of offspring, the timing of reproduction, age and size at maturity and growth pattern, longevity) [6]. These studies have shown that temporal fluctuations in magnitude and direction of directional selection are common [7] and that the magnitude of directional selection is sufficient to produce rapid microevolutionary changes in many populations.
Directional natural selection is forecasted to increase with more frequent droughts and rising temperatures [8].
Despite all the potential benefits of mixtures and evolutionary populations, for many years the only example of practical exploitation remained the one on malting barley mixtures, which spread in the former German Democratic Republic up to 360,000 hectares with a reduction of the percentage of fields affected by severe mildew epidemics from 50 to 10% and a threefold reduction of the percentage of fields sprayed with fungicides [9].
The major obstacle to translating into practice the several findings of research about the benefits of mixtures and evolutionary populations was likely the seed legislation, which in several countries is based on the principles of variety registration and seed certification. Seed can be marketed only if it belongs to a variety that has been registered and the seed has been certified. A variety must satisfy distinctness, uniformity and stability requirements (DUS) [10].
International recognitions
The European Union, recognizing the advantages of evolutionary populations and mixtures, in the past 10 years, invested considerably on research in diversity in agriculture, first with the project “Strategies for Organic and Low-Input Integrated Breeding and Management” (SOLIBAM) from 2010 to 2014, then with the project “Embedding Crop Diversity and Networking for Local High Quality Food Systems (DIVERSIFOOD) from 2015 to 2019, more recently with Boosting Organic Seed and Plant Breeding across Europe (LIVESEED), and eventually with the new EU “farm to fork” strategy.
In 2014, the European Commission, recognizing the contradiction between funding research project to promote the use of diversity in agriculture and the seed laws that hinder it, issued an “Implementing decision” (https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX%3A32014D0150) with which a “temporary experiment at Union level” was organized “for the purpose of assessing whether the production, with a view to marketing and marketing, under certain conditions, of seed from populations belonging to the species Oats, Wheat, Barley, and Corn, may constitute an improved alternative to the exclusion of the marketing of seed not complying with the requirements…..”. The experiment initially planned to end on 31st December 2018, was eventually extended to 28 February 2021.
Furthermore, in 2018, the European Commission adopted a new Regulation {(EU) 2018/848, https://eur-lex.europa.eu/legal-content/EN/ALL/?uri=CELEX%3A32018R0848}, which lays down new rules on the controls of organic production and labelling of organic products, and which make available for use in organic production plant reproductive material that does not belong to a variety, but rather belongs to a plant grouping within a single botanical taxon with a high level of genetic and phenotypic diversity between individual reproductive units. In the Regulation this material is defined as heterogenous material, which: a) presents common phenotypic characteristics; b) is characterised by a high level of genetic and phenotypic diversity between individual reproductive units, so that the plant grouping is represented by the material as a whole, and not by a small number of units; c) is not a variety within the meaning of Article 5(2) of Council Regulation (EC) No 2100/94 (1); d) is not a mixture of varieties; and e) has been produced in accordance with this Regulation.
In 2018, the International Fund for Agricultural Development (IFAD), based in Rome, funded a project “Using Agricultural Biodiversity and Farmers’ Knowledge to Adapt Crops to Climate Change in Iran” with which evolutionary plant breeding on wheat and barley was initiated in Iran by the International NGO CENESTA [11]. As a follow-up, IFAD funded in 2018 a four-years project “Use of genetic diversity and Evolutionary Plant Breeding for enhanced farmer resilience to climate change, sustainable crop productivity, and nutrition under rainfed conditions”, implemented by Bioversity International and involving Uganda and Ethiopia in Africa, Jordan and Iran in the Near East and Nepal and Bhutan in the Far East. As part of this project, a number of evolutionary populations and mixtures of wheat, barley, rice and beans are tested in several villages in the six countries with the participation of farmers.
References
- Kremen C, Miles A. 2012. Ecosystem Services in Biologically Diversified versus Conventional Farming Systems: Benefits, Externalities, and Trade-Offs. Ecology and Society 17(4): 40.
- Ceccarelli S, Grando S. 2022. Evolutionary Plant Breeding with an Introduction to Participatory Plant Breeding, The Alliance of Bioversity International and CIAT, Rome pp 125 (in press).
- Ceccarelli S, Grando S, Maatougui M, Michael M, Slash M, Haghparast R, Rahmanian M, Taheri A, Al-Yassin A, Benbelkacem A, Labdi M, Mimoun M, Nachit M. 2010. Plant Breeding and Climate Changes. Journal of Agricultural Science 148(6): 627 – 638.
- Murphy KM, Carter AH, Jones SS. 2013. Evolutionary Breeding and Climate Change. In: Kole C (eds) Genomics and Breeding for Climate-Resilient Crops. Springer, Berlin, Germany, pp 377 – 389.
- Ceccarelli S, Grando S. 2020. Evolutionary plant breeding as a response to the complexity of climate change. IScience 23 (12): 101815.
- Kingsolver JG, Hoekstra HE, Hoekstra JM, Berrigan D, Vignieri SN, Hill CE, Hoang A, Gibert P, Beerli P. 2001. The Strength of Phenotypic Selection in Natural Populations. The American Naturalist 157: 245 – 261.
- Siepielski AM, Di Battista JD, Carlson SM. 2009. It’s about time: the temporal dynamics of phenotypic selection in the wild. Ecology Letters 12 (11): 1261 – 1276.
- Exposito-Alonso M, 500 Genomes Field Experiment Team, Burbano HA, Bossdorf O, Nielsen R, Weigel D. 2019. Natural selection on the Arabidopsis thaliana genome in present and future climates. Nature 573 (7772): 126 – 129.
- Wolfe MS, Brändle U, Koller B, Limpert E, McDermott JM, Müller K, Schaffner D. 1992. Barley mildew in Europe: population biology and host resistance. Euphytica 63 (1–2), 125 – 139.
- Winge T. 2015. Seed Legislation in Europe and Crop Genetic Diversity. In: Lichtfouse E. (eds) Sustainable Agriculture Reviews. Sustainable Agriculture Reviews, vol 15. Springer, Cham. Switzerland.
- Ceccarelli S, Grando S, Salimi M, Razawi K. 2022. Evolutionary Populations for Sustainable Food Security and Food Sovereignty. In (eds Y. Nishikawa and M. Pimbert) Seeds for Diversity and Inclusion. Agroecology and endogenous development. Springer Nature Switzerland AG Gewerbestrasse 11, 6330 Cham, Switzerland.
Ricevi i nuovi contenuti direttamente nella tua casella di posta.
